|
Bronze typically consists of 88% copper and 12% tin.This is prevented by adding small impurities to white tin. White tin will transform into gray tin when the temperature falls below 13.2 degrees C.Other elements in pewter generally include copper, antimony, and bismuth. Comprehensive data on the chemical element Tin is provided on this page including scores of properties, element names in many languages, most known nuclides of. Pewter is a tin alloy that is at least 85% tin.This is due to the breaking of the crystal structure of the atoms. When a bar of tin is bent, it will make a screaming sound called a "tin cry".This is the most stable isotopes of all the elements. The symbol "Sn" comes from the Latin word for tin, "stannum." Tin gets its name from the Anglo-Saxon language. Bronze was harder than pure copper and was easier to work with and cast. Tin was first heavily used starting with the Bronze Age when tin was combined with copper to make the alloy bronze. Tin has been known about since ancient times. Other applications for tin include metal alloys such as bronze and pewter, the production of glass using the Pilkington process, toothpaste, and in the manufacture of textiles. Tin cans are actually steel cans covered with a plating of tin. Tin is also used as a plating to protect other metals such as lead, zinc, and steel from corrosion. Solder is a mixture of tin and lead that is used to join pipes and to make electronic circuits. The majority of tin today is used to make solder. There are estimates that the minable tin on Earth will be gone in 20 to 40 years. The majority of tin is mined in China, Malaysia, Peru, and Indonesia. It is around the 50th most abundant element in the Earth's crust. It is generally not found in its free form. Tin is found in the Earth's crust primarily in the ore cassiterite. This allows it to be used as a plating material to protect other metals. Tin is resistive to corrosion from water. 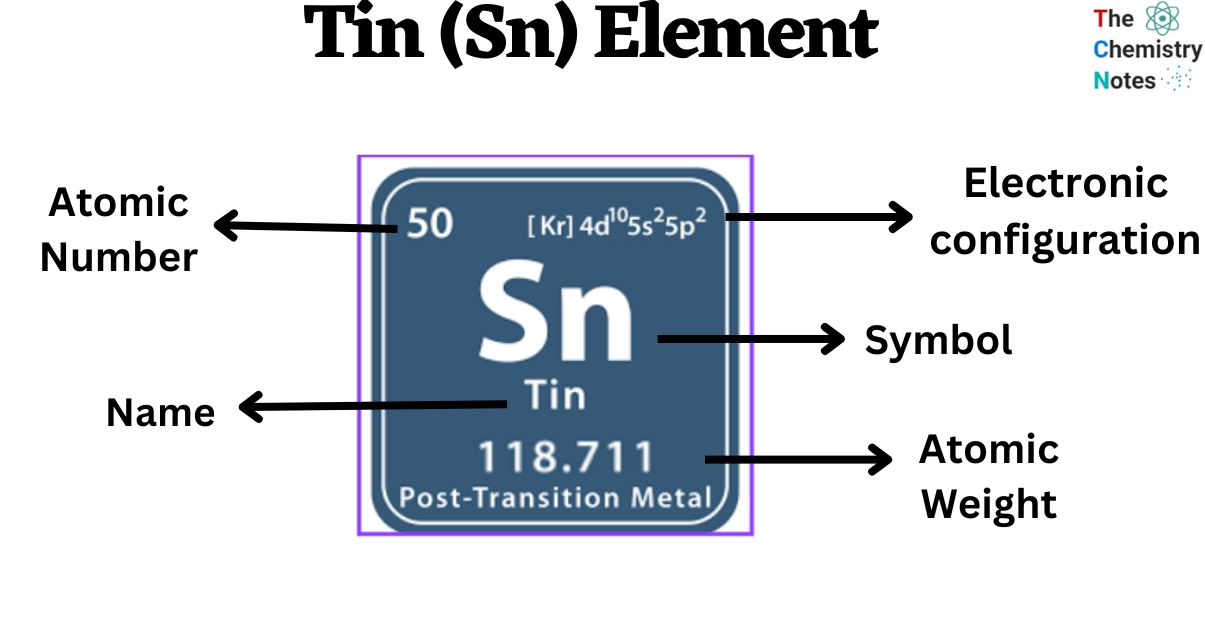
Gray tin is non-metallic and is a gray powdery material. White tin is the metallic form of tin we are most familiar with. Tin can form two different allotropes under normal pressure. It is very malleable (meaning that it can be pounded into a thin sheet) and can be polished to a shine. Under standard conditions tin is a soft silvery-gray metal. Tin atoms have 50 electrons and 50 protons with 4 valence electrons in the outer shell. If you’d like to see a periodic table in Spanish, check out this Periodic Table, or Tabla Peridica de los Elementos in Spanish. It is classified as a post-transition metal. Different languages have different words for the same thing, why should the elements be any different This table is a list of the elements in English and Spanish arranged by atomic number. Tin is the fourth element of the fourteenth column of the periodic table. Discovered by: Known about since ancient times.Density (white): 7.365 grams per cm cubed.Sometimes there may be multiple compounds with that element on one side, so you'll need to use your best judgement and be prepared to go back and try the other options. Step 2: Multiply coefficients for compounds to balance out each elementįor each element that is not equal, try to balance it by adding more of it to the side with less. Step 1: Count the number of each element on the left and right hand sides When using the inspection method (also known as the trial-and-error method), this principle is used to balance one element at a time until both sides are equal and the chemical equation is balanced. To be balanced, every element in Sn + HCl = SnCl2 + H2 must have the same number of atoms on each side of the equation. A chemical element, often simply called an element, is a type of atom which has the same number of protons in its atomic nucleus (i.e., the same atomic number, or Z ). The law of conservation of mass states that matter cannot be created or destroyed, which means there must be the same number atoms at the end of a chemical reaction as at the beginning. This is a list of the 118 chemical elements that have been identified as of 2023.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
